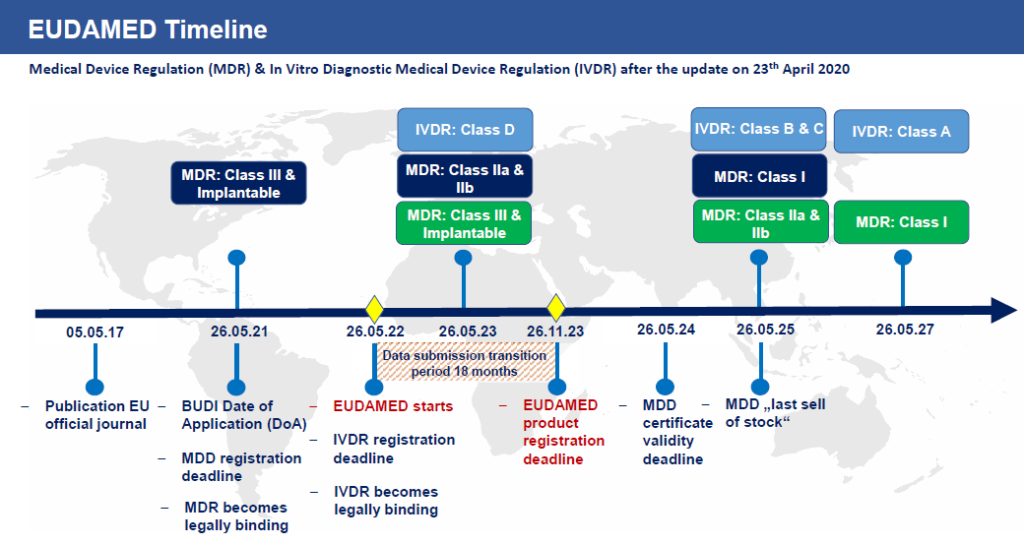
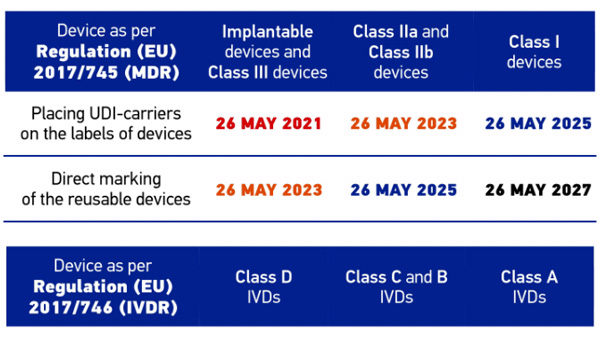
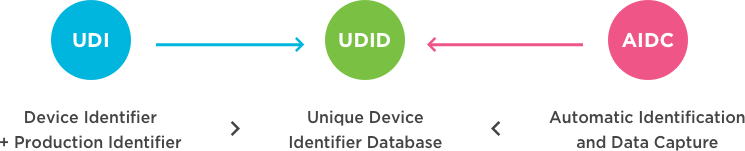
UDI Beginners Guide: Unique Device Identification (EU MDR and IVDR) - Medical Device Regulation and ISO quality standard

European Union Medical Device Regulation and In Vitro Device Regulation: unique device identification | BSI
Medical devices- UDI regulation - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup