
BLOG: What are the new device classifications in the In vitro Diagnostic Regulation (IVDR)? | RegMetrics

New European In Vitro Diagnostic Medical Devices Regulation - BDR | Blackhills Diagnostics Resources

Tips and tools for dealing with the in-vitro diagnostics regulation (IVDR) | European Cluster Collaboration Platform

TÜV SÜD @TUVSUD emite el primer certificado con el reglamento IVDR a un producto clase B – Enhorabuena!!

The In vitro diagnostic medical devices regulation (EU) 2017/746: what will change for public health laboratory services?

The In vitro diagnostic medical devices regulation (EU) 2017/746: what will change for public health

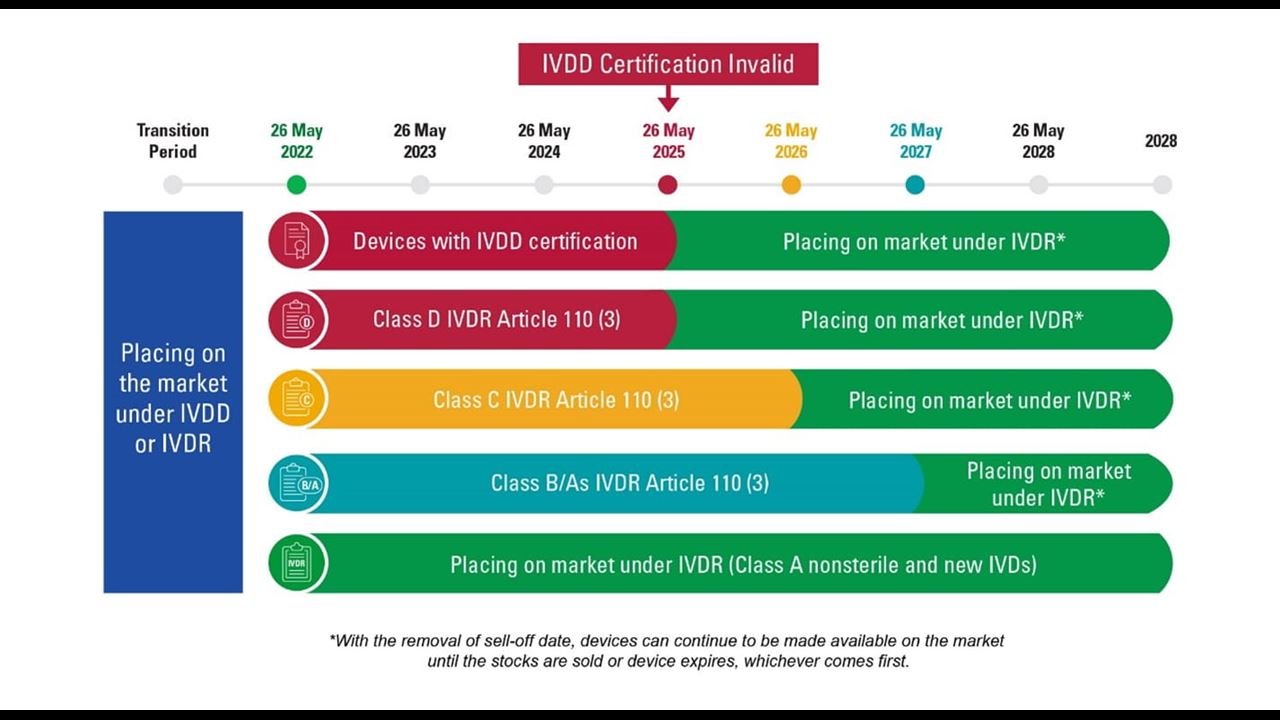
.png)


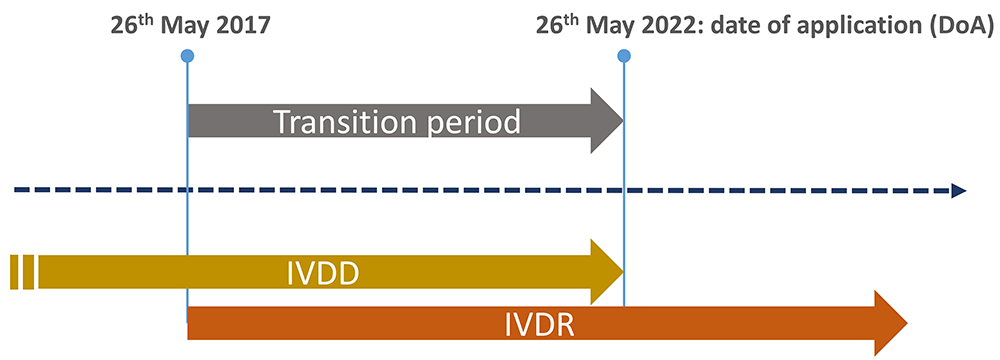







/tuv-rheinland-ivdr-visual-2-en_core_1_x.png)




/tuv-rheinland-ivdr-visual-1-en_core_1_x.png)