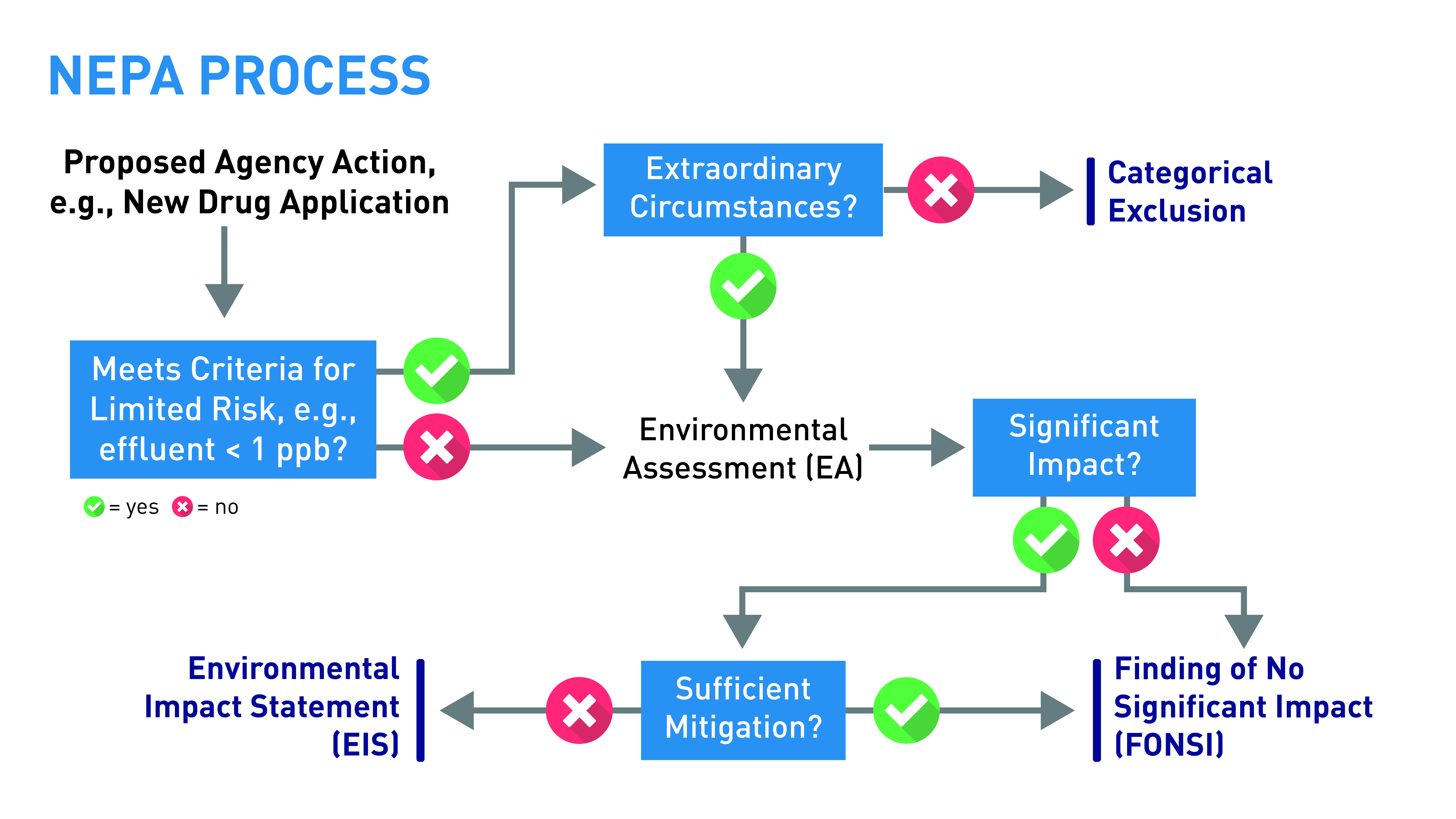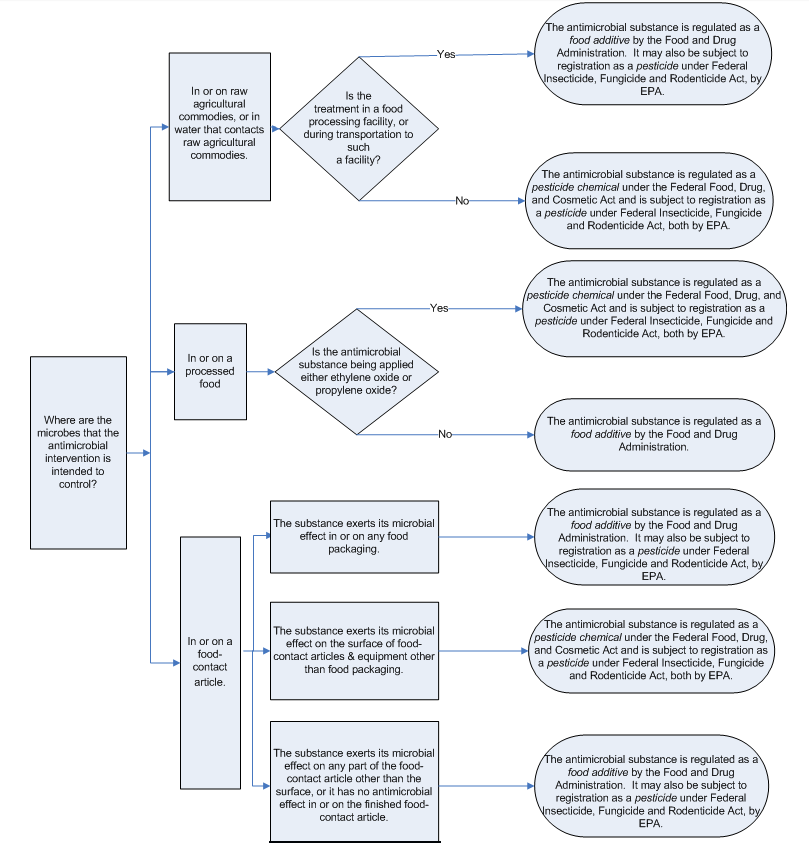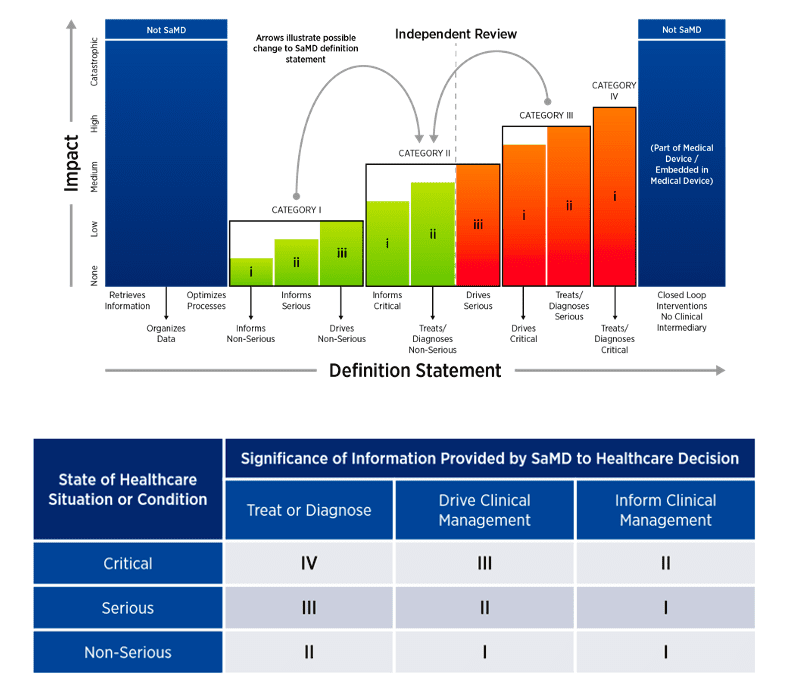
Software as a medical device: Here's how the regulatory landscape is changing - Medical Design and Outsourcing

PPT - An Overview of the United States Regulatory Environment (FDA and Healthcare) for Global Pharmaceutical Compliance Profe PowerPoint Presentation - ID:1286593

FDA Regulatory Environment for Wearables, Implantables, and Other Digital Health Devices - Wearables Crash Course Webinar Series - Epstein Becker & Green, P.C.

Nanomaterials in Consumer Products and FDA Regulation George A. Kimbrell The International Center for Technology Assessment Nano & Bio In Society Conference. - ppt download