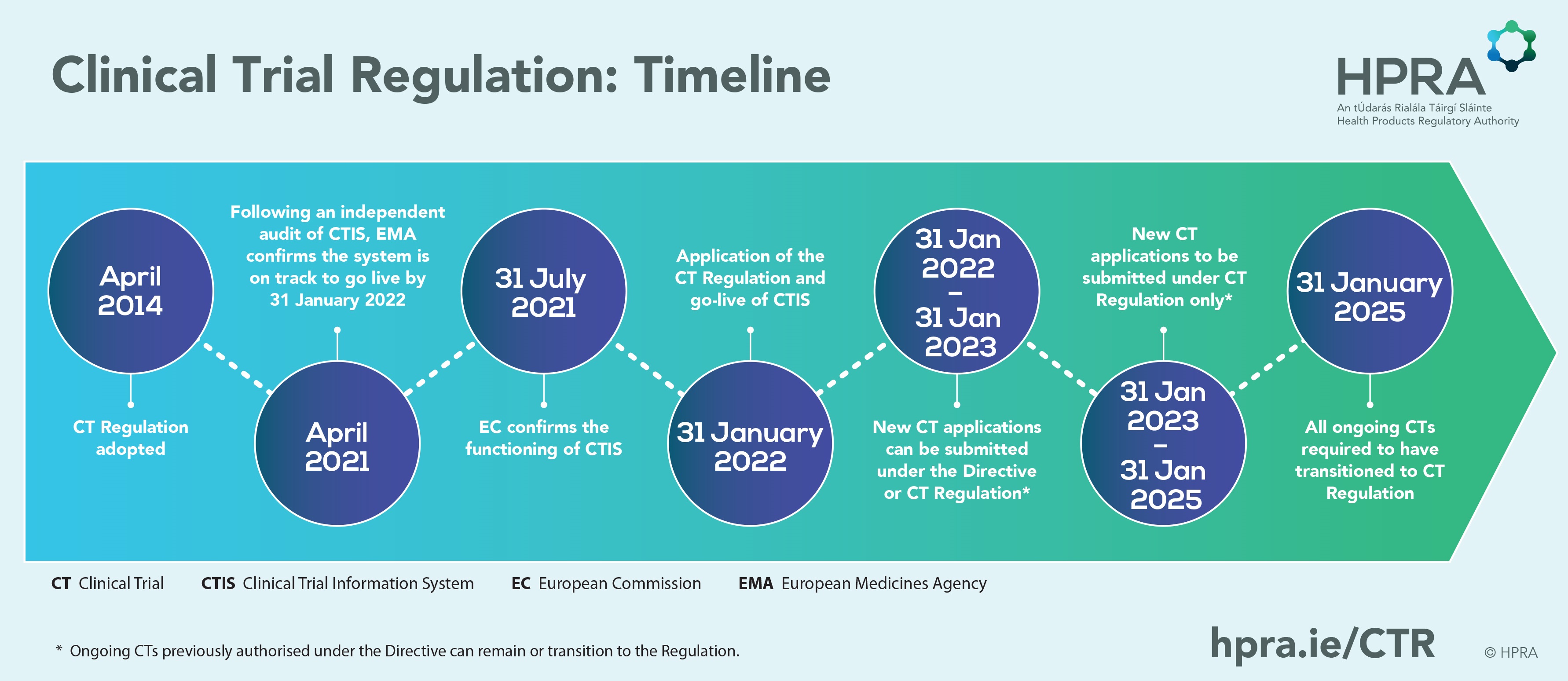
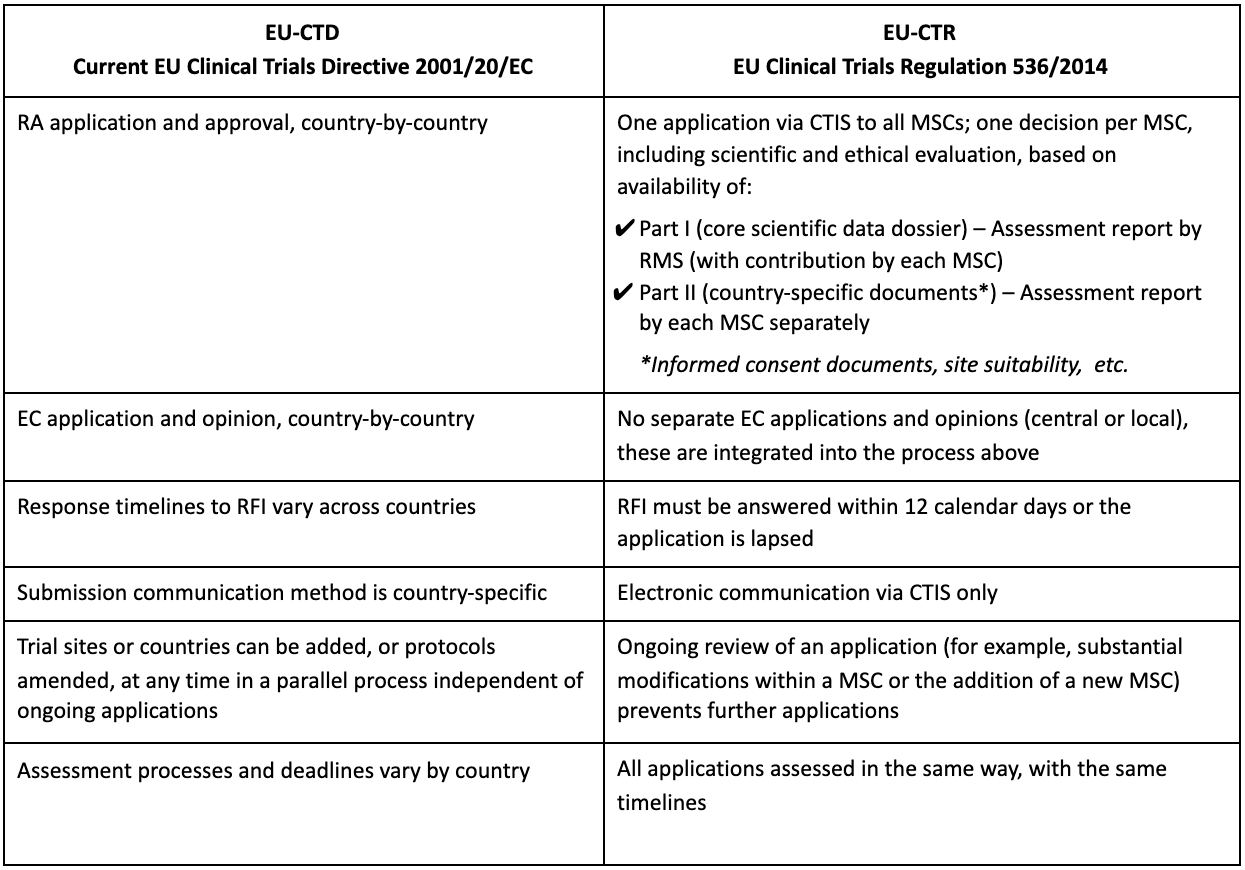
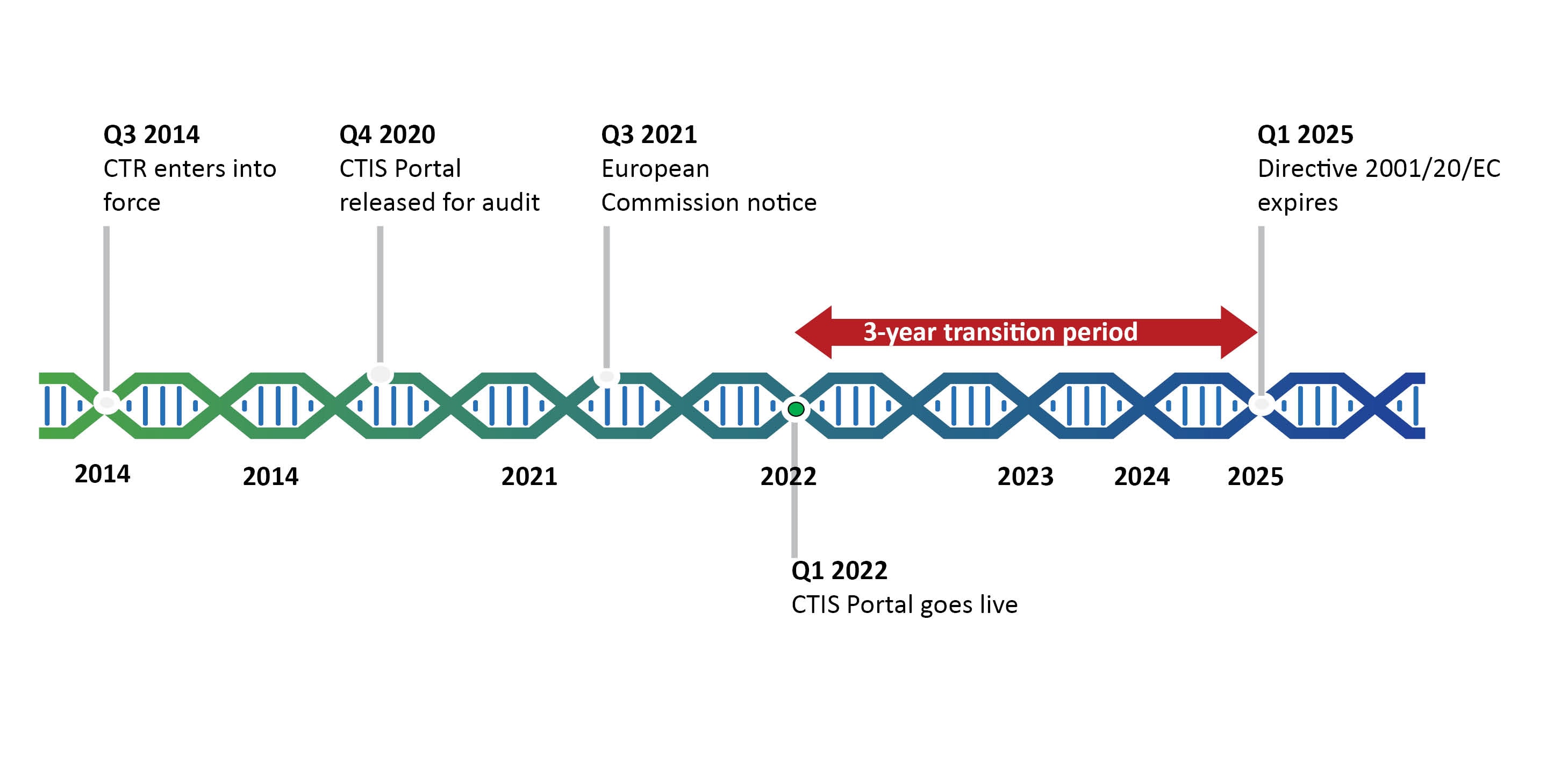
GCP and Quality in “Regulation (EU) 536/2014 on clinical trials on medicinal products for human use and repealing Directive 2001/20/EU” - ScienceDirect
Presentation - Key features and objectives of Regulation EU No 536 /014 - What is new, what has changed? (Laura Pioppo)